mesenchymal stem cells near me sets the stage for this fascinating topic, offering readers a glimpse into a story that explores their potential therapeutic applications in regenerative medicine.
The discovery of mesenchymal stem cells has revolutionized the field of regenerative medicine, providing a new hope for treating various degenerative disorders. With their ability to differentiate into multiple cell types and modulate the immune system, mesenchymal stem cells have shown great promise in treating conditions such as Parkinson’s disease, osteoarthritis, and multiple sclerosis. Through their unique properties, mesenchymal stem cells have the potential to repair damaged tissues and organs, making them an exciting area of research in the field of medicine.
Understanding the Concept of Mesenchymal Stem Cells and Their Therapeutic Potential in Regenerative Medicine: Mesenchymal Stem Cells Near Me

The term “regenerative medicine” emerged a few decades ago as medical science advanced rapidly. This discipline aims to repair, replace, or regenerate cells, tissues, or organs to maintain or restore normal function. Regenerative medicine has become a crucial area, offering innovative treatments for diseases and injuries that traditional medicine cannot solve. Among these approaches, mesenchymal stem cells (MSCs) stand out as key players in various regenerative processes.
Mesenchymal stem cells were first identified in 1970 by Friedenstein and his colleagues in a study involving bone marrow cells. The researchers isolated a group of cells capable of differentiating into various bone and cartilage cells. This groundbreaking discovery paved the way for extensive research on MSCs, which has since unraveled their potential in treating various degenerative disorders.
The Historical Background of Mesenchymal Stem Cells
The discovery of MSCs marked a significant milestone in the field of regenerative medicine. Over the years, researchers have extensively studied their properties, behavior, and therapeutic potential. The early studies focused on their ability to differentiate into cells of the mesenchyme, a type of tissue that gives rise to bone, cartilage, and connective tissue. These findings sparked interest in MSCs as a promising tool for treating tissue damage and degenerative diseases.
Understanding the Therapeutic Potential of Mesenchymal Stem Cells
Mesenchymal stem cells possess unique properties that make them an attractive candidate for regenerative medicine. These cells are known for their ability to:
* Differentiate into various cell types, including bone, cartilage, and fat cells.
* Produce growth factors that support tissue repair and regeneration.
* Modulate the immune system to reduce inflammation and promote healing.
* Engraft in tissues, where they can survive and function for extended periods.
These characteristics make MSCs a valuable asset in the pursuit of regenerative medicine.
Examples of Mesenchymal Stem Cells’ Application in Treating Degenerative Disorders
Researchers have investigated the potential of MSCs in treating various degenerative disorders, including:
- Osteoarthritis: MSCs have been shown to differentiate into cartilage cells, promoting cartilage repair and regeneration.
- Spinal cord injury: MSCs have been found to support neural tissue repair and improve functional recovery.
- Rheumatoid arthritis: MSCs have been shown to modulate the immune system and reduce inflammation, leading to improved symptoms and quality of life.
Conclusion
The discovery of mesenchymal stem cells has revolutionized the field of regenerative medicine. Their unique properties and therapeutic potential make them an exciting area of research. As we continue to uncover the secrets of MSCs, we may unlock new treatments for degenerative disorders, improving the lives of countless individuals.
Overview of Mesenchymal Stem Cell Therapy and Its Current Status in Medical Practice
Mesenchymal stem cell (MSC) therapy has emerged as a groundbreaking approach in regenerative medicine, offering immense potential for treating various diseases and injuries. The ability of MSCs to home and engraft in target tissues, promoting tissue repair and regeneration, has sparked significant interest in the medical community.
Mesenchymal stem cell therapy has shown remarkable progress in recent years, with numerous studies demonstrating its efficacy in treating a range of conditions, including autoimmune diseases, cardiovascular disorders, and musculoskeletal injuries. The mechanisms underlying MSC homing and engraftment are complex, involving interactions between MSCs and the target tissue microenvironment.
Mechanisms of Mesenchymal Stem Cell Homing and Engraftment
Mesenchymal stem cells utilize various mechanisms to home and engraft in target tissues, including:
- Cell-cell interactions: MSCs interact with various cell types, including endothelial cells, pericytes, and immune cells, to establish a symbiotic relationship with the target tissue.
- Cytokine and chemokine signaling: MSCs secrete a range of cytokines and chemokines that help them home and engraft in target tissues, promoting tissue repair and regeneration.
- Homotypic adhesion molecules: MSCs express homotypic adhesion molecules, such as integrins and selectins, that facilitate their adhesion and migration to target tissues.
These mechanisms enable MSCs to adapt to the local tissue microenvironment, promoting tissue repair and regeneration.
Different Delivery Methods of Mesenchymal Stem Cells for Therapeutic Purposes
Mesenchymal stem cells can be delivered via various routes for therapeutic purposes, including:
- Via injection: MSCs can be injected into the target tissue through a needle, allowing for localized treatment.
- Via intravenous administration: MSCs can be administered intravenously, where they can home to target tissues and promote tissue repair and regeneration.
- Via gene therapy: MSCs can be engineered to express therapeutic genes, allowing for targeted treatment of diseases.
- Via tissue engineering: MSCs can be used to create tissue-engineered constructs, such as scaffolds and matrices, to promote tissue repair and regeneration.
Each delivery method has its advantages and limitations, and the choice of delivery method depends on the specific disease or injury being treated, as well as the patient’s overall health and condition.
Investigating Potential Biomarkers for Predicting Mesenchymal Stem Cell Therapy Outcomes in Patients
Accurately assessing the safety and efficacy of mesenchymal stem cell-based treatments in clinical trials poses a significant challenge. With the increasing interest in leveraging mesenchymal stem cells for regenerative medicine, identifying reliable biomarkers that can predict treatment outcomes assumes even greater importance. In this context, researchers seek to develop experimental models that can pinpoint biomarkers capable of forecasting the success of mesenchymal stem cell therapies.
Challenges in Assessing Safety and Efficacy
Several factors contribute to the complexity of evaluating mesenchymal stem cell treatments in clinical trials. Firstly, the inherent variability within stem cell populations, including differences in donor characteristics and cell isolation procedures, introduces an element of unpredictability. Additionally, the multifaceted nature of regenerative medicine makes it challenging to account for the complex interplay between the stem cells, the host environment, and the disease condition being targeted. Consequently, standardization of clinical trial protocols and endpoint assessment becomes crucial.
Experimental Models for Identifying Biomarkers
The development of suitable experimental models is crucial for predicting mesenchymal stem cell therapy outcomes. Several approaches have been explored, including animal models and in vitro systems. For instance, mouse models have been used to investigate the efficacy of mesenchymal stem cell transplantation in repairing damaged tissues. Similarly, in vitro studies employing primary cell cultures have enabled researchers to evaluate the behavior of mesenchymal stem cells in controlled settings.
Key Experimental Models
- The use of immunocompromised mouse models allows for the assessment of mesenchymal stem cell integration and survival within the host environment.
- Animal models of autoimmune diseases, such as rheumatoid arthritis and multiple sclerosis, have been employed to investigate the therapeutic potential of mesenchymal stem cells in modulating immune responses.
- Cell culture models using mesenchymal stem cells and co-cultured immune cells can provide insights into the mechanisms underlying immune regulation.
Identifying Reliable Biomarkers
Biomarker identification in the context of mesenchymal stem cell therapies requires careful consideration of the biological processes influenced by these cells. Several potential biomarkers have been identified, including:
‘Cysteine-rich angiogenic inducer 61 (Cyr61)’, a secreted protein involved in angiogenesis and tissue repair.
‘Vascular endothelial growth factor (VEGF)’, a key regulator of angiogenesis, which has been shown to promote mesenchymal stem cell-induced vascularization.
Implications for Clinical Studies
The identification of reliable biomarkers that can predict mesenchymal stem cell therapy outcomes will significantly enhance the translation of these treatments to the clinical setting. This will enable healthcare providers to optimize treatment strategies and improve patient prognoses. Moreover, biomarker-based monitoring of treatment efficacy will facilitate the timely identification of responders versus non-responders, allowing for tailored therapeutic interventions.
Understanding the Impact of Mesenchymal Stem Cells on the Immunological Environment and Their Potential to Treat Autoimmune Disorders
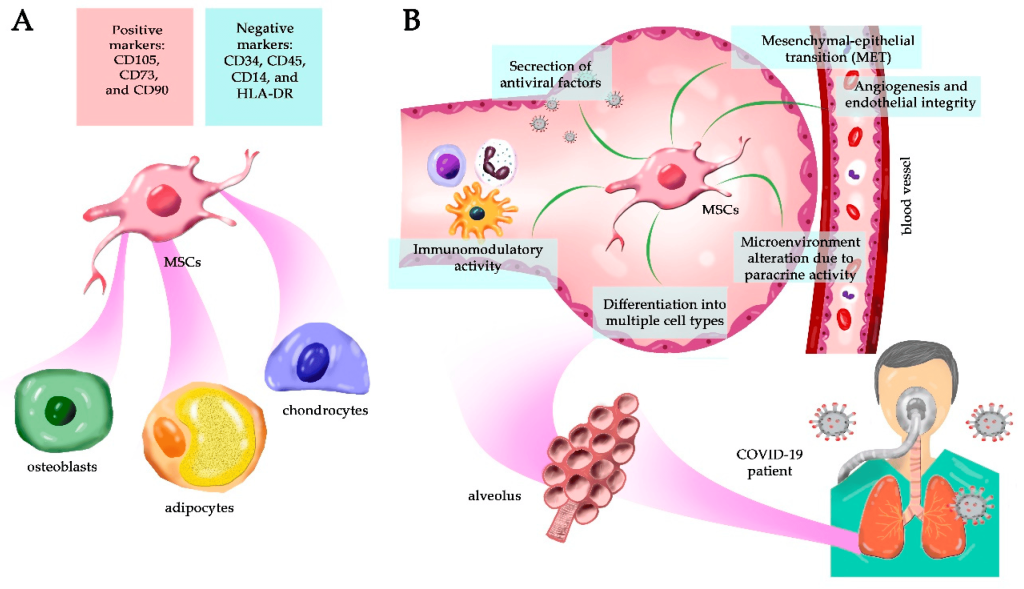
Mesenchymal stem cells (MSCs) have garnered significant attention in recent years for their potential to treat autoimmune disorders. These cells possess unique properties that allow them to interact with the immune system, modulate its response, and potentially restore balance to the immunological environment. In this context, we will delve into the immune suppressive properties of MSCs and their potential to treat various autoimmune diseases.
The Immune Suppressive Properties of Mesenchymal Stem Cells
MSCs have been shown to exhibit immunosuppressive properties through various mechanisms, including the secretion of anti-inflammatory cytokines, the modulation of immune cell function, and the induction of regulatory T cells. One of the key factors underlying these properties is the ability of MSCs to migrate to sites of inflammation, where they can interact with immune cells and modulate their response. This process is facilitated by the expression of specific surface molecules, such as CD44 and CD90, which allow MSCs to recognize and bind to their target cells.
Comparison of Therapeutic Effects in Different Autoimmune Diseases
While MSCs have shown promise in treating various autoimmune diseases, the effectiveness of this approach can vary depending on the specific condition being targeted. For example, studies have demonstrated that MSCs can alleviate symptoms and improve outcomes in patients with rheumatoid arthritis, multiple sclerosis, and systemic lupus erythematosus. In contrast, the efficacy of MSCs in treating conditions such as type 1 diabetes has been more limited, with some studies suggesting that MSCs may even exacerbate the disease in certain cases.
Underlying Molecular Mechanisms
The molecular mechanisms underlying the immunosuppressive properties of MSCs involve a complex interplay of signaling pathways, cytokines, and surface molecules. Some of the key molecules involved in this process include:
- TGF-β (transforming growth factor beta): This cytokine has been shown to induce the expression of anti-inflammatory genes in immune cells and promote the differentiation of regulatory T cells.
- IL-10 (interleukin-10): This cytokine has been implicated in the downregulation of pro-inflammatory cytokines and the promotion of tissue repair.
- CD52: This surface molecule has been shown to play a role in the modulation of immune cell function and the induction of regulatory T cells.
These molecules, along with others, contribute to the immunosuppressive properties of MSCs and their potential to treat autoimmune diseases.
Regulatory T Cells and MSCs
Regulatory T cells (Tregs) play a crucial role in maintaining immune tolerance and preventing autoimmune disease. MSCs have been shown to induce the differentiation of Tregs, which can then promote immune tolerance and reduce inflammation. The interaction between MSCs and Tregs is complex and involves a range of cytokines and surface molecules.
The induction of Tregs by MSCs is thought to be mediated by the secretion of TGF-β and IL-10, which can downregulate pro-inflammatory cytokines and promote the differentiation of Tregs.
This process highlights the potential of MSCs to modulate the immune response and restore balance to the immunological environment.
Challenges and Future Directions
While MSCs have shown promise in treating autoimmune diseases, several challenges must be addressed before this approach can be widely adopted. These challenges include:
- The need for standardized protocols for MSC isolation and expansion.
- The development of more efficient delivery methods for MSCs.
- The elucidation of the molecular mechanisms underlying the therapeutic effects of MSCs.
Addressing these challenges will require further research and collaboration among scientists, clinicians, and industry partners.
Overview of the Current Regulatory Framework governing Mesenchymal Stem Cell Research and Therapy
The regulatory landscape surrounding mesenchymal stem cell research and therapy is complex and constantly evolving. As these cells hold great promise for regenerative medicine, governments and regulatory agencies worldwide are working to establish frameworks that balance the need for innovation with the requirement for safety and efficacy. This overview aims to provide a comprehensive understanding of the current regulatory landscape and the challenges associated with translating mesenchymal stem cell research into therapeutic products.
The Global Regulatory Landscape
The global regulatory landscape for mesenchymal stem cell therapy varies significantly across countries and regions. While some countries have established well-defined regulatory pathways, others are still in the process of developing their frameworks. This complexity poses significant challenges for researchers, clinicians, and industry stakeholders seeking to bring mesenchymal stem cell therapies to market. Despite these challenges, many countries are actively working to establish clear guidelines and regulations to facilitate the development of these therapies.
Regulatory Frameworks for Mesenchymal Stem Cell Therapy in Various Countries or Regions
The following table provides an overview of the regulatory frameworks for mesenchymal stem cell therapy in various countries or regions:
| Country/Region | Regulatory Agency | Key Regulatory Requirements |
|---|---|---|
| United States | US FDA | Cell therapy regulations under GMP, Investigational New Drug (IND) application, and biologics license application |
| European Union | European Medicines Agency (EMA) | Advanced therapy medicinal product (ATMP) regulation, clinical trial authorization, and marketing authorization |
| Japan | Japanese Ministry of Health, Labour and Welfare (MHLW) | Cell therapy regulations under GMP, pharmaceuticals and medical devices act |
| China | China Food and Drug Administration (CFDA) | Cell therapy regulations under GMP, pharmaceuticals and medical devices regulations |
Challenges Associated with Translating Mesenchymal Stem Cell Research into Therapeutic Products
Despite the growing body of evidence supporting the therapeutic potential of mesenchymal stem cells, several challenges must be addressed before these cells can be brought to market. Some of the key challenges include:
- Lack of standardization in cell isolation, expansion, and banking procedures
- Inadequate understanding of cell biology and mechanisms of action
- Inconsistent preclinical and clinical trial data
- Divergent regulatory frameworks and requirements across countries and regions
Future Directions for Regulatory Frameworks, Mesenchymal stem cells near me
As mesenchymal stem cell research and therapy continue to evolve, regulatory frameworks must adapt to address emerging challenges and opportunities. Some potential future directions for regulatory frameworks include:
- Development of standardized guidelines for cell isolation, expansion, and banking procedures
- Investment in basic research to better understand cell biology and mechanisms of action
- Enhanced collaboration and coordination across countries and regions to facilitate global development of mesenchymal stem cell therapies
Investigating the Efficacy of Mesenchymal Stem Cells in Treating Cancer-Related Complications and Side Effects
In the realm of regenerative medicine, mesenchymal stem cells (MSCs) have emerged as a promising tool for addressing various medical concerns, including cancer-related complications and side effects. Cancer treatments such as chemotherapy and radiation therapy can cause damage to healthy tissues and organs, leading to debilitating symptoms and quality-of-life impairments. As researchers delve deeper into the therapeutic potential of MSCs, their ability to modulate the tumor microenvironment has garnered significant attention. This narrative explores the tumor microenvironment and the roles of MSCs within this context, followed by a discussion of the current evidence for their efficacy in treating cancer-related complications and side effects.
The tumor microenvironment is a complex network of cells, tissues, and molecules that interact to facilitate tumor growth and progression. It is composed of various cell types, including cancer cells, immune cells, endothelial cells, and fibroblasts, each contributing to the tumor’s ability to evade immune surveillance, adapt to changing environments, and metastasize. One of the key roles of MSCs within this environment is to modulate the interactions between these cell types.
Modulation of the Tumor Microenvironment by MSCs
MSCs have been shown to interact with the tumor microenvironment in various ways, including modulating the immune response, promoting angiogenesis, and suppressing tumor growth. Research has demonstrated that MSCs can inhibit the function of immune cells, such as T lymphocytes and natural killer cells, which are essential for recognizing and eliminating cancer cells. This immune suppression can facilitate tumor growth and progression, highlighting the potential risks associated with using MSCs for cancer therapy.
In contrast, some studies have suggested that MSCs can also promote anti-tumor immune responses by stimulating the activation and expansion of immune cells, such as dendritic cells and T lymphocytes. By presenting antigens and releasing cytokines and chemokines, MSCs can create an environment that supports immune surveillance and tumor elimination. This dual role of MSCs in modulating the tumor microenvironment underscores the complexity of their interactions with the tumor and highlights the need for further research to fully understand their effects.
Current Evidence for the Efficacy of MSCs in Treating Cancer-Related Complications and Side Effects
Despite the challenges and controversies surrounding the use of MSCs for cancer therapy, research has demonstrated potential benefits in treating cancer-related complications and side effects. For instance, MSC-based therapies have shown promise in reducing inflammation, improving tissue regeneration, and minimizing organ damage associated with cancer treatments. These effects can lead to improved quality of life for patients undergoing cancer therapy and may even enhance their overall survival rates.
Some notable studies have investigated the use of MSCs for specific cancer-related complications, such as radiation-induced bone marrow damage and chemotherapy-induced peripheral neuropathy. These studies have reported promising results, including enhanced bone marrow recovery, reduced neuropathy symptoms, and improved functional outcomes. While further research is needed to confirm the efficacy and safety of MSC-based therapies, these findings offer hope for the development of innovative treatments for cancer-related complications and side effects.
In conclusion, the role of MSCs in modulating the tumor microenvironment and their potential benefits in treating cancer-related complications and side effects warrant continued investigation. Further research is necessary to fully understand the interactions between MSCs and the tumor microenvironment, as well as the risks and benefits associated with their use in cancer therapy. By exploring the therapeutic potential of MSCs, researchers and clinicians may develop innovative treatments that alleviate the debilitating symptoms and quality-of-life impairments associated with cancer and its treatments.
- MSCs can modulate the tumor microenvironment by interacting with various cell types, including cancer cells, immune cells, and endothelial cells.
- MSCs can promote anti-tumor immune responses by stimulating the activation and expansion of immune cells, such as dendritic cells and T lymphocytes.
- Research has demonstrated potential benefits of MSC-based therapies in treating cancer-related complications and side effects, including inflammation, tissue regeneration, and organ damage.
| Cancer Treatment | Cancer-Related Complication | MSC-Based Therapy Effects |
|---|---|---|
| Radiation therapy | Bone marrow damage | Enhanced bone marrow recovery, reduced organ failure |
| Chemotherapy | Peripheral neuropathy | Reduced neuropathy symptoms, improved functional outcomes |
MSCs have the potential to revolutionize the treatment of cancer-related complications and side effects, offering novel avenues for enhancing patient quality of life and survival rates.
Examining the Potential of Mesenchymal Stem Cells as a Tool for Gene Therapy and Genetic Diseases
Mesenchymal stem cells (MSCs) have garnered significant attention in recent years for their potential in treating various genetic diseases and conditions. Their unique ability to differentiate into multiple cell types, including neural, muscle, and bone cells, makes them an attractive candidate for gene therapy. In this section, we will delve into the mechanisms by which MSCs can be engineered for gene therapy and discuss the current status of gene therapy research using MSCs.
Engineered Mesenchymal Stem Cells for Gene Therapy
Engineered MSCs can be designed to express specific therapeutic genes or to edit existing genes to correct genetic deficiencies. This can be achieved through various methods, including gene transfer, gene editing, and cell fusion. Gene transfer involves the introduction of a therapeutic gene into MSCs, which can then express the gene product. Gene editing techniques, such as CRISPR-Cas9, enable the precise modification of genes within MSCs. Cell fusion involves the combination of MSCs with other cells that have the desired therapeutic properties.
- Gene Transfer: The most common method of gene transfer is viral-mediated transduction, which involves the use of viral vectors to introduce the therapeutic gene into MSCs.
- Gene Editing: CRISPR-Cas9 and other gene editing tools enable precise gene editing within MSCs, allowing for the correction of genetic mutations associated with various diseases.
- Cell Fusion: This technique involves the fusion of MSCs with other cells, such as immune cells or stem cells, to create hybrid cells with the desired therapeutic properties.
Current Status of Gene Therapy Research Using Mesenchymal Stem Cells
Gene therapy research using MSCs is an active area of investigation, with numerous preclinical and clinical studies ongoing. These studies aim to evaluate the safety and efficacy of MSC-based gene therapy for various genetic diseases, including sickle cell anemia, muscular dystrophy, and Parkinson’s disease. Although results have been promising, challenges associated with MSC-based gene therapy, such as low gene transfer efficiency and immune rejection, need to be addressed.
“The use of MSCs for gene therapy offers a promising approach for treating genetic diseases, but further research is needed to optimize the efficacy and safety of these therapies.”
In conclusion, mesenchymal stem cells have the potential to revolutionize gene therapy and the treatment of genetic diseases. Engineered MSCs can be designed to express specific therapeutic genes or to edit existing genes to correct genetic deficiencies. Gene therapy research using MSCs is an active area of investigation, with promising results from preclinical and clinical studies. However, challenges associated with MSC-based gene therapy need to be addressed to ensure the safe and effective use of these therapies.
Concluding Remarks
In conclusion, mesenchymal stem cells near me have the potential to revolutionize the field of regenerative medicine, providing new hope for treating various degenerative disorders. As research continues to advance, we can expect to see more breakthroughs in the use of mesenchymal stem cells for therapeutic purposes. With their unique properties and potential therapeutic applications, mesenchymal stem cells are an exciting area of research that holds great promise for the future.
Q&A
What are mesenchymal stem cells?
Mesenchymal stem cells are a type of adult stem cell that can differentiate into multiple cell types, including bone, cartilage, muscle, and fat cells.
How are mesenchymal stem cells used in regenerative medicine?
Mesenchymal stem cells are used to repair damaged tissues and organs, and to modulate the immune system in order to treat various degenerative disorders.
Are mesenchymal stem cells safe?
Mesenchymal stem cells have been shown to be safe and well-tolerated in clinical trials, but further research is needed to fully understand their potential risks and benefits.
Can mesenchymal stem cells be used to treat cancer?
Mesenchymal stem cells may have the potential to be used to treat cancer by targeting cancer stem cells and modulating the tumor microenvironment, but further research is needed to fully understand their potential anticancer effects.